-
3 - items
-
 APOTEX Corp., CEFTRIAXONE SODIUM POWDER INJECTION SDV 1GM/VL , Part # 60505614804 , 10/BX, “This Item Requires Pharmaceutical License to Place an Order”
1 x
$22.50
APOTEX Corp., CEFTRIAXONE SODIUM POWDER INJECTION SDV 1GM/VL , Part # 60505614804 , 10/BX, “This Item Requires Pharmaceutical License to Place an Order”
1 x
$22.50
-
 WATER BACTERIOSTATIC 30ML FOR INJECTION MULTIDOSE VIAL , HOSPIRA WORLDWIDE, INC, Part # 00409397703, 25/BX “This Item Requires Pharmaceutical License to Place an Order”
1 x
$269.00
WATER BACTERIOSTATIC 30ML FOR INJECTION MULTIDOSE VIAL , HOSPIRA WORLDWIDE, INC, Part # 00409397703, 25/BX “This Item Requires Pharmaceutical License to Place an Order”
1 x
$269.00
-
 BD #1727170107 freeflex® Sodium Chloride 0.9% IV Solution 1,000 mL 10Bags/Case MOQ 1000 Cases
1 x
$300.00
BD #1727170107 freeflex® Sodium Chloride 0.9% IV Solution 1,000 mL 10Bags/Case MOQ 1000 Cases
1 x
$300.00
- View Cart
-
GRIFOLS 0.9% Sodium Chloride Injection – 500ML in Fleboflex® plastic container – 20Bags/CS – Minimum Order Qty *1000 Cases*
$500.00
Description
0.9% Sodium Chloride Injection, USP, is a sterile, nonpyrogenic solution for fluid and electrolyte replenishment in Fleboflex® plastic container for intravenous administration.
Fleboflex® is a flexible container free of PVC, plasticizers, adhesives, and latex
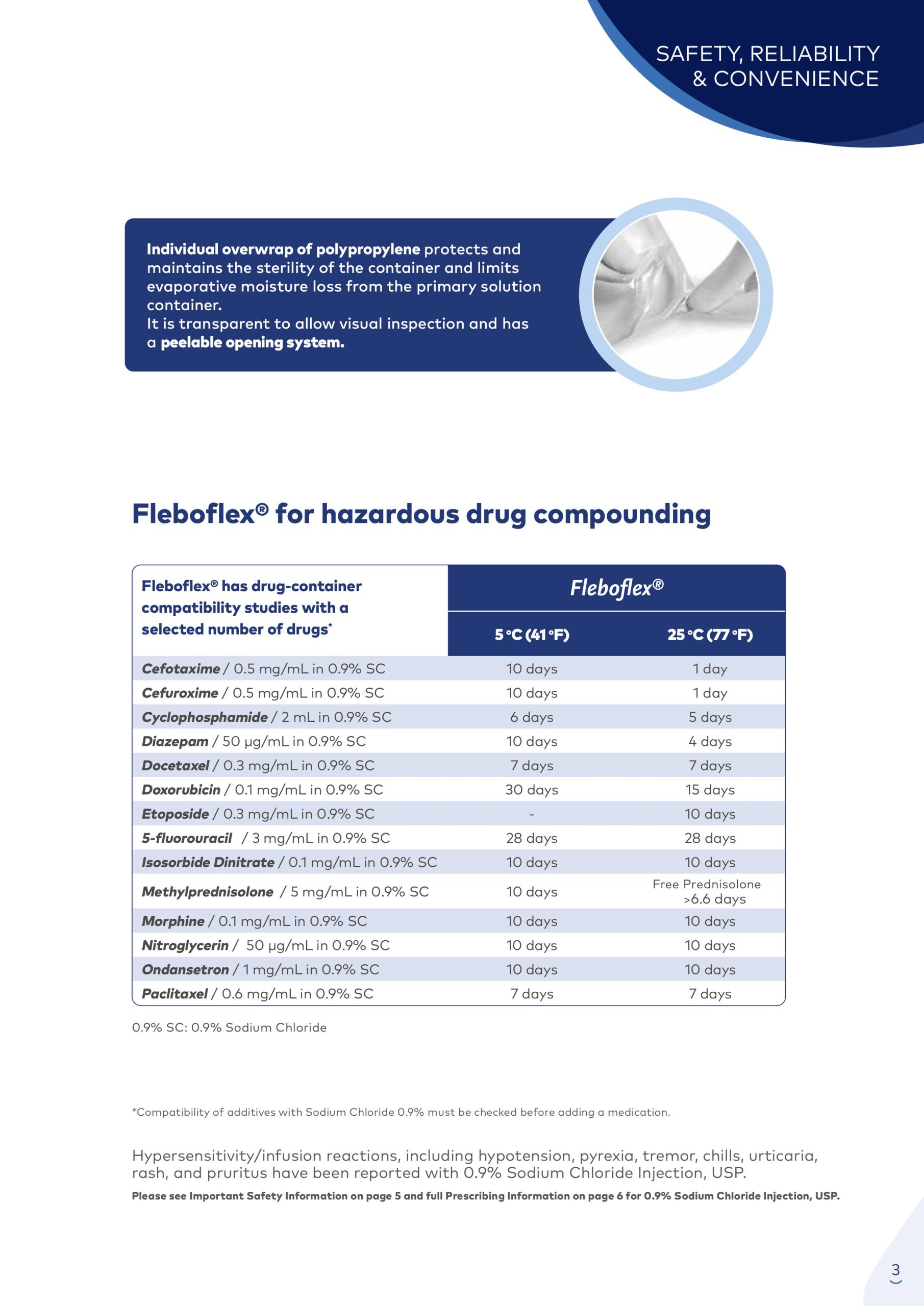
The Fleboflex® container consists of a polypropylene multilayer film. Polypropylene is a highly compatible material. It is used for the preparation of intravenous mixtures with drugs that have shown their incompatibility with other plastics
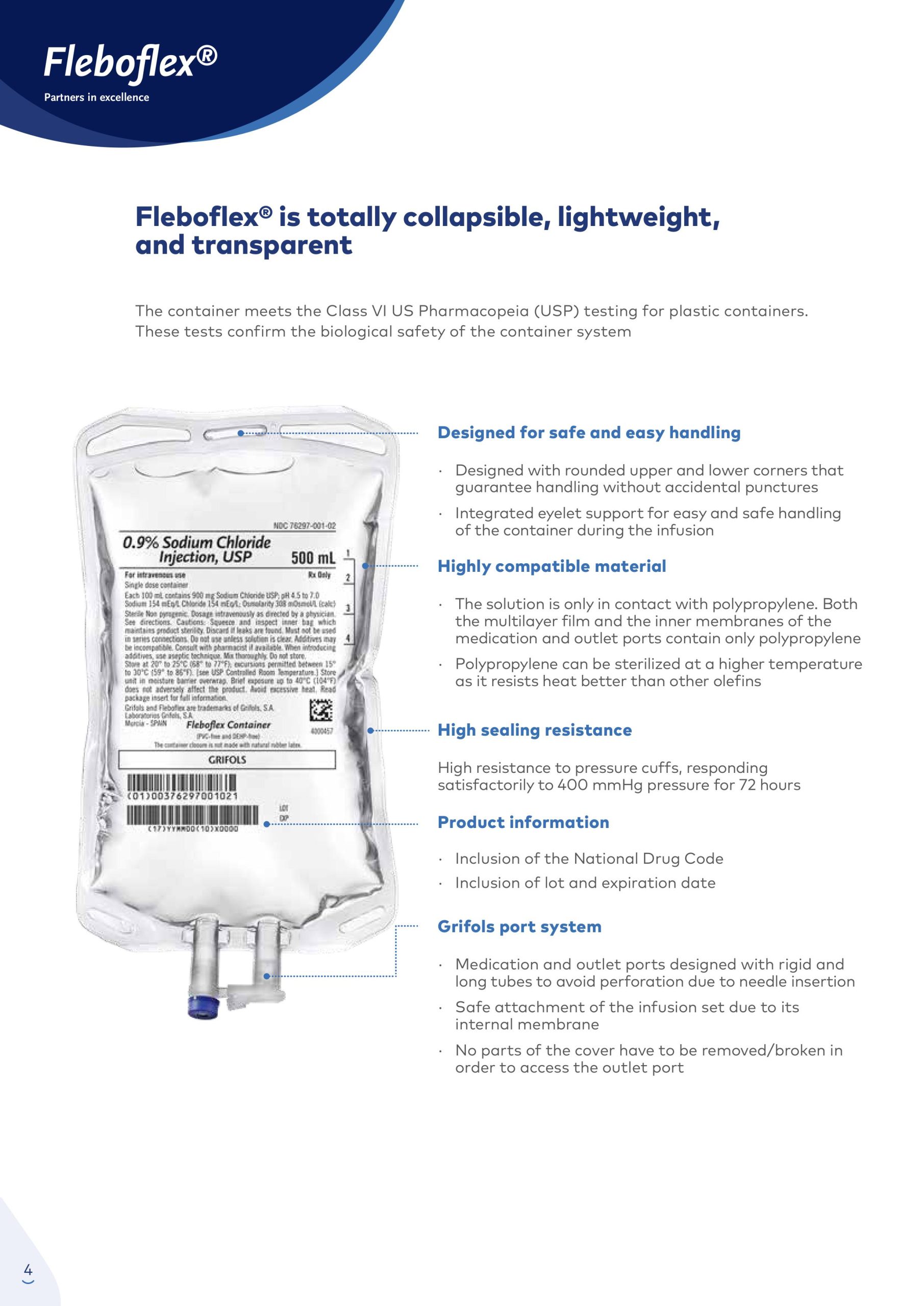
Fleboflex® is totally collapsible, lightweight, and transparent
The Fleboflex® container meets the Class VI US Pharmacopeia (USP) testing for plastic containers. These tests confirm the biological safety of the container system